 The stability of this water-in-water emulsion from coalescence is attributed to three molecular forces:ġ. 
To minimize the overall energy of the system, the DSCG molecules in the droplet prefer to align either parallel or perpendicular to the surfaces of the droplets.(Fig. As water solvated droplets in a w/w emulsion, DSCG molecules would align in a preferred direction on the surface of the droplet. Surprisingly, some of these water-in-water emulsions can be exceptionally stable from coalescence for up to 30 days.īecause molecules of liquid crystal assume a preferred common orientation among themselves, the overall orientation of liquid crystals in a droplet is only stable in certain configurations (Fig. Consequently, the known polymers that afford w/w emulsion include polyacrylic amides and polyols. As such, ionic polymer when mixed with DSCG does not form w/w emulsion, but gives rise to a homogeneous solution or a precipitate solution. Apart from being water-soluble, one important criterion for the generation of this w/w emulsion system is that the polymer cannot bear functional groups that interact strongly with DSCG. The polymer solution serves as the medium or continuous phase of the w/w emulsion. Thus, the separation of hydrophobic/ hydrophilic groups cannot be applied to DSCG. Unlike conventional lyotropic liquid crystals which consist of oily molecules such as 5CB, DSCG molecules are not amphiphilic, but entirely water-soluble. This molecule is an anti-asthmatic drug, but also exists as a special type of liquid crystal when the concentration of DSCG is ~9-21 wt%. The liquid crystal component of the emulsion is disodium cromolyn glycate (DSCG). This water-in-water emulsion consists of liquid crystals suspended as water-solvated droplets dispersed in a solution of polymer whose solvent is also water. Structure of disodium cromolyn glycate, DSCG This w/w emulsion was generated when the different water-solvated molecular functional groups get segregated in an aqueous mixture consisting of polymer and liquid crystal molecules. These molecular interactions include hydrogen bonding, pi stacking, and salt bridging. Recently, such a water-in-water emulsion was demonstrated to exist and be stable from coalescence by the separation of different types of non- amphiphilic, but water-soluble molecular interactions. As such, when two entirely aqueous solutions containing different water-soluble molecules are mixed, water droplets containing predominantly one component are dispersed in water solution containing another component. Water-in-water (W/W) emulsion is a system that consists of droplets of water-solvated molecules in another continuous aqueous solution both the droplet and continuous phases contain different molecules that are entirely water-soluble. JSTOR ( June 2014) ( Learn how and when to remove this template message). 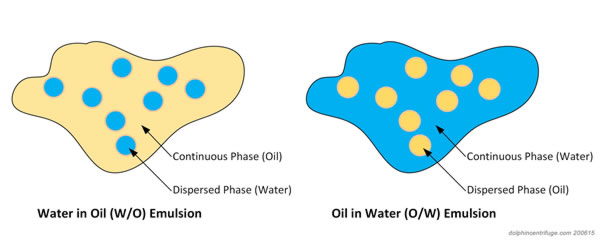
Unsourced material may be challenged and removed.įind sources: "Water-in-water emulsion" – news Please help improve this article by adding citations to reliable sources. Oil-in-water emulsions are non-greasy and easily removable from the skin surface while water-in-oil emulsions are greasy and not water washable.This article needs additional citations for verification. In an oil-in-water (o/w) emulsions, the dispersed phase (discontinuous or internal phase) phase is oil, and the dispersion medium (continuous or external phase) is water while in a water-in-oil (w/o) emulsions the water is the dispersed phase, and oil the dispersion medium.Ģ. Differences between oil-in-water emulsion and water-in-oil emulsionġ. This article focuses on the differences between oil-in-water emulsion and water-in-oil emulsion. Pharmaceutical emulsions are currently used internally for the administration of nutrients, drugs, and diagnostic agents. Very frequently emulsions are used in cosmetic products as topical vehicle for dermal application since they have high patient/consumer acceptance. Emulsions have been widely used in many areas of application: in industries, agriculture, food technologies, pharmaceutics, and cosmetics. An emulsion is a dispersion of at least two immiscible liquids, one of which is dispersed as droplets in the other liquid, and stabilized by an emulsifying agent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
